Article

According to the American Cancer Society™ nearly half of the American male population (40.14%) will contract some type of invasive cancer in their lifetime. What’s more, 1 in 5 (21.34%) will die from cancer. The statistics for the American female population aren’t much better, with 38.70% likely to contract an invasive form of cancer, and 1 in 5 (18.33%) likely to die.
These sobering numbers help highlight what’s at stake in the fight against cancer and how crucial oncology studies are in this battle.
Oncology is “the branch of medicine that deals with the study and treatment of cancer.” If you search clinicaltrials.gov, a registry of federally and privately supported clinical trials conducted both in the U.S. and around the globe, there are currently 39,506 different studies for Neoplasm, Cancer, and Tumor listed.
In 2015, the Pharmaceutical Research and Manufacturers of America (PhRMA) published research on industry-sponsored clinical trials, including oncology studies. They found that “oncology accounted for both the largest number of clinical trials (2,560 trials, or more than 40% of industry-sponsored trials), and the largest number of trial participants (215,176, or nearly 19% of participants).”
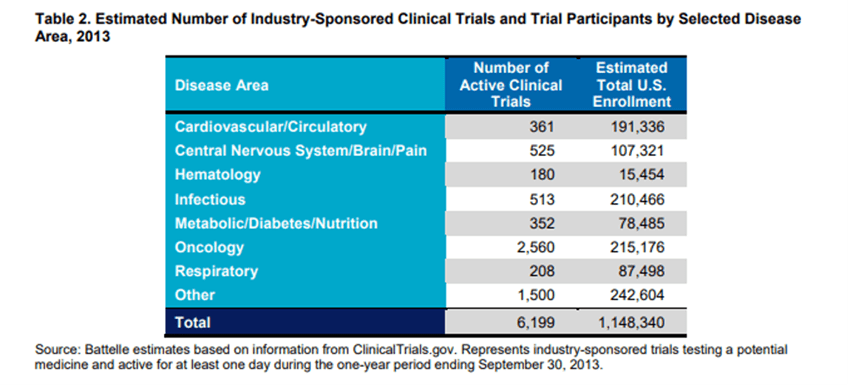
Additionally, oncology trials showed the highest average per-patient cost, with an average per-patient cost of $59,500. This is due in part to the fact that oncology trials typically require fewer participants than other types of clinical trials, meaning any fixed costs are spread out over a smaller number of participants. And back in 2006, developing and marketing a new cancer drug was estimated to be around $1 billion.
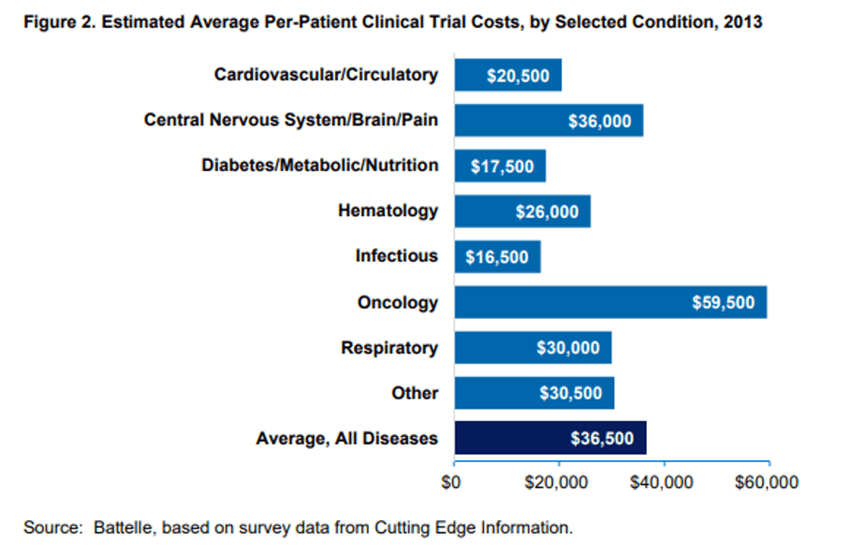
Altogether this data showcases the sheer size and cost of oncology clinical trials. But the challenges don’t stop there.
It has been repeatedly estimated that less than 5% of adult cancer patients even enroll in oncology clinical trials. However, an estimated 70% of Americans are eager and willing to participate in clinical trials. The paper, Role of Clinical Trial Participation in Cancer Research: Barriers, Evidence, and Strategies, theorizes that structural (e.g. access to cancer clinics, insurance), clinical (e.g. eligibility to participate), and attitudinal barriers (both via physicians and patients) create the large gap between trial participant rates and the willingness of patients to participate in such trials.
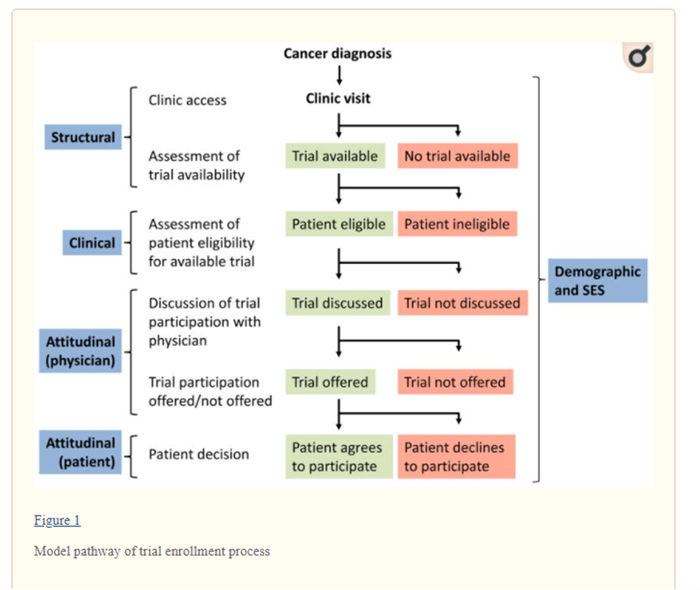
Overall, the success of oncology clinical trials greatly depends on the level of engagement of its participants, oncologists, clinical staff, and patients from the very beginning. However, continuous barriers and roadblocks, such as the ones briefly described above, create both logistically and psychologically difficult scenarios that often discourage otherwise willing participants.
Anticipating potential barriers and roadblocks before they happen is key to the success of your oncology trial. To help you on your journey, we encourage you to consider these important questions:
Is the designated site properly equipped and staffed to support the needs of the trial?
Is it easy for trial participants to access the trial site? Does secondary transportation need to be arranged?
Are the hired staff knowledgeable and prepared to properly conduct your clinical trial?
Have eligible patients been assessed for fear and discomfort in regards to taking part in the trial? Is counseling available? Have relevant risks been made known?
Is the provider reliable? Can they deliver kits to multiple sites on a global scale? Could there be any supply chain issues that impact sourcing materials? What’s the expected shelf life for your kits?
Read More: Key Considerations for Clinical Trial Specimen Collection
Do you have a solution to easily access and harmonize your data across global sites? What happens if trial timelines become extended? Are you aware of regulatory complexities impacting costs? Did you plan for the possibility of Increased cost and availability of trial supplies and equipment? How about rising courier costs?
Read More: Managing the Cost of Your Clinical Trial
Do they have robust global capabilities? Are they experienced with oncology trials? Can they provide personalized service? Will they deliver on time and on budget? Will your study be a priority?
Read More: Selecting a Central Lab to Support the Clinical Trial Process
The inability to answer these types of questions poses potential setbacks to the success of your oncology clinical trial — opening your trial up to delays, headaches, and cost increases. At ACM, we routinely help CROs and Sponsors for oncology clinical studies consider these questions as their central laboratory partner of choice. We anticipate barriers and roadblocks for them and apply creative solutions to mitigate the impact, resulting in successful clinical studies.
Learn About Our Central Lab Services
As the need for oncology studies continues to grow, the potential for delivering life-saving and sustaining treatments and therapies that bring health, healing, and hope to patients and their loved ones grows with it.
“Year over year we’ve seen an increase in the number of oncology clinical trials at ACM Global Laboratories," said Andrey Fesyuk, Supervisor of Clinical Trials Anatomic Pathology. "Our team offers expertise with rigorous data collection and reporting requirements for pathology and cytology testing. We will continue to deliver actionable clinical insights that lead to new treatments and therapies that bring health, healing, and hope to those diagnosed with cancer and the ones who love them.”
When it comes to clinical trials — experience matters. This is especially true of oncology trials, which play such a huge part in the clinical trial landscape. At ACM Global Laboratory, we’re proud to have been involved with more than 57 oncology clinical trials in the past 5 years alone.
We know every clinical trial is unique, and that’s why we work with you to deliver a personalized, white-glove touch to exactly fit your needs. ACM’s team of laboratory professionals span all areas of clinical expertise including pathology, flow cytometry, specialized biomarkers and pharmacogenomics. We also have robust teams for study and specimen management, IT services and more.
Contact us today to discuss how ACM can help make your oncology clinical trial a success.